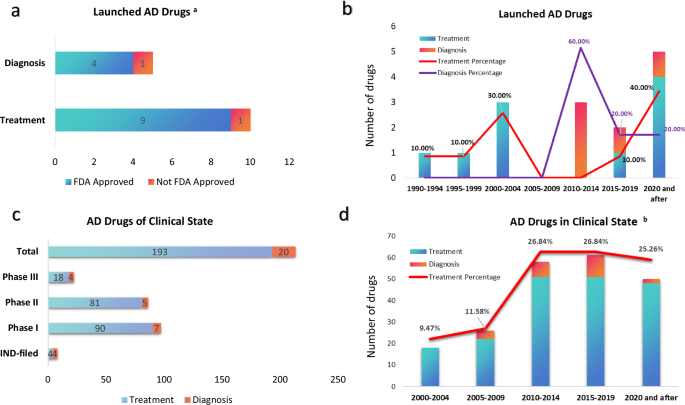
For decades, the fight against Alzheimer's disease was marked by a heartbreaking pattern of high hopes and clinical setbacks. The scientific community, guided by early discoveries around amyloid-beta proteins, pursued treatments that ultimately could not alter the disease's grim trajectory in patients. This cycle of progress and disappointment led to a long, quiet period in therapeutic development, leaving families and clinicians with limited options. Yet, that landscape is now transforming with palpable momentum, driven by relentless scientific inquiry and biotechnology advances. A new wave of discovery has begun, cresting with the full FDA approval of lecanemab, the first genuine disease-modifying therapy in twenty years, signaling a turning point from managing symptoms to addressing the underlying biology of Alzheimer's itself.
The current pipeline for Alzheimer's therapies is more robust and diverse than ever before. While the recent approvals of antibodies like lecanemab and donanemab, which target amyloid plaques, represent a monumental validation of long-held research paths, they are just the beginning. An analysis of the clinical landscape reveals over two hundred drugs in various stages of development, with a significant portion exploring targets beyond amyloid. Inflammation and immune system pathways are now receiving nearly equal attention, reflecting a broadening understanding of the complex mechanisms at play in the brain. This diversification includes investigations into tau protein tangles, cellular signaling, and even novel vaccine approaches, painting a picture of a field no longer reliant on a single hypothesis but attacking the problem from multiple, innovative angles.
This flourishing research ecosystem thrives on a vital partnership between agile biotech pioneers and established pharmaceutical leaders. The early, high-risk stages of discovery are predominantly fueled by small and medium-sized enterprises, where novel concepts and cutting-edge approaches take shape. These entities serve as the innovation engine, exploring differentiated targets and advanced strategies. As promising candidates advance, larger firms increasingly provide the critical funding and extensive clinical trial expertise necessary to shepherd them through late-stage development and to patients. This synergistic model ensures a balanced pipeline, combining groundbreaking creativity with the resources required for validation and global access, ultimately bringing new hope from the lab to the clinic.
The impact of this progress is already extending beyond therapeutics into the crucial realm of diagnosis. During periods when treatment development seemed stagnant, research into diagnostic tools quietly accelerated, leading to several new approvals. These advances are key, as earlier and more accurate detection allows for interventions at stages where emerging disease-modifying treatments may be most effective. Together, the expansion of both diagnostic capabilities and the therapeutic arsenal marks a profound shift. For the millions of individuals and families navigating an Alzheimer's diagnosis, the journey is increasingly illuminated by genuine optimism. The path forward is one of continued refinement, combination approaches, and persistent discovery, moving steadily toward a future where Alzheimer's disease can be effectively managed and, one day, prevented.