A long-term agreement between a medical device company and a major research reactor will ensure the scalable production of an innovative liver cancer therapy as it moves toward potential commercialization.
The partnership names the University of Missouri Research Reactor (MURR) as the primary irradiation partner for Eye90 microspheres, a radioembolization treatment for hepatocellular carcinoma. The formalized alliance builds on seven years of prior collaboration that has supported the therapy's development and the ongoing Route90 FDA IDE clinical trial, which is now fully enrolled. This step is designed to create a reliable supply chain for the treatment as it advances beyond clinical testing.
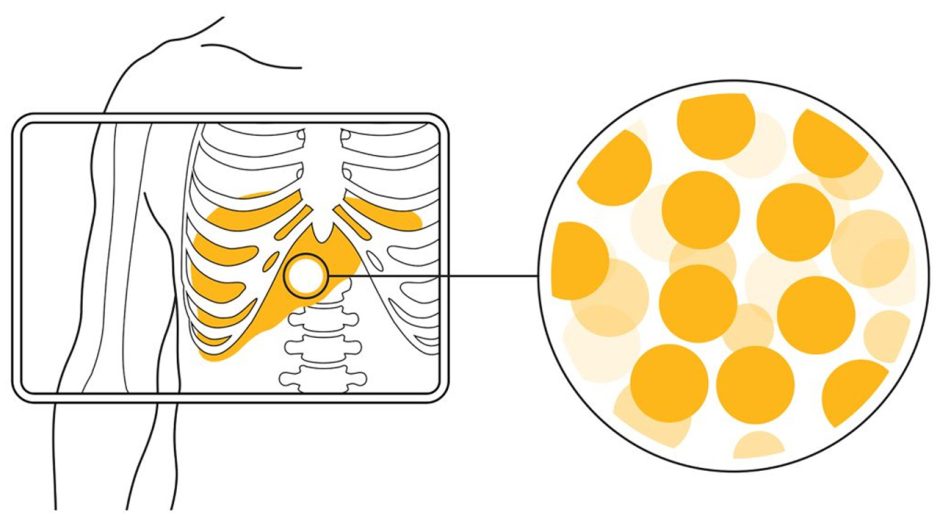
The therapy works by delivering millions of tiny radioactive spheres directly into the blood vessels feeding a liver tumor. The spheres, irradiated with Yttrium-90 at MURR, become lodged and deliver a high dose of radiation to the cancer while largely sparing healthy tissue. This targeted approach, known as selective internal radiation therapy, is a growing area of oncology. MURR is currently the only location in the United States producing Y-90 for both clinical and commercial use, making it a critical infrastructure for this and other nuclear medicine treatments.
Officials from both institutions highlighted the partnership's broader impact. They stated that combining ABK's device technology with MURR's isotope production expertise aims to accelerate global patient access to the therapy. Furthermore, the collaboration is framed as a boost to Missouri's economy and its standing in medical innovation, keeping high-tech research and production within the state.
The next phase hinges on the results of the pivotal Route90 trial and subsequent regulatory processes. With a secured production pathway now in place, the partners are positioning Eye90 microspheres to move more swiftly from research to clinical practice, offering a hopeful outlook for patients with liver cancer worldwide.